NEWS
Researchers Shed Further Light onto Zinc Homeostasis in Cells
A research group has unearthed how zinc transporter complexes regulate zinc ion (Zn2+) concentrations in different areas of the Golgi apparatus and revealed that this mechanism finely tunes the chaperone protein ERp44.
The findings, which were reported in the journal Nature Communications on May 9, 2023, reveal the crucial chemical and cellular biological mechanism at play behind zinc homeostasis, something necessary for avoiding fatal diseases such as diabetes, cancers, growth failures, and immunodeficiency.
As a trace element, zinc is essential for our health. Zn2+ are vital for enzyme catalysis, protein folding, DNA binding, and regulating gene expression, with nearly 10% of human proteome binding Zn2+ for their structural maturation and function.
Secretory proteins like hormones, immunoglobulins, and blood clotting factors are synthesized and folded in the endoplasmic reticulum (ER), a complex membrane network of tubules. Subsequently, they are transported to and matured in the Golgi apparatus, the organelle composed of multiple flattened sacs called cisternae, which sorts and processes proteins before directing them to a specific destination. Chaperone proteins are vital for maintaining protein homeostasis and preventing the formation of misfolded or aggregated proteins in these organelles.
The group's previous research demonstrated that Zn2+ in the Golgi apparatus plays an essential part in protein quality control in the early secretory pathway comprising the ER and Golgi. This system is mediated by the ER-Golgi cycling chaperone protein ERp44.
In the Golgi apparatus, there exists three ZnT complexes: ZnT4, Znt5/6, and ZnT7. Yet, until now, mechanisms of how Zn2+ homeostasis is maintained in the Golgi apparatus has remained unclear.
"Using chemical biology and cell biology approaches together, we revealed that these ZnT complexes regulate the Zn2+ concentrations in the different Golgi compartments, namely cis, medial, and trans-Golgi cisternae," says Kenji Inaba, a corresponding author of the study and professor at Tohoku University's Institute of Multidisciplinary Research for Advanced Materials Sciences. "We also further elucidated the intracellular transport, localization, and function of ERp44 controlled by ZnT complexes."
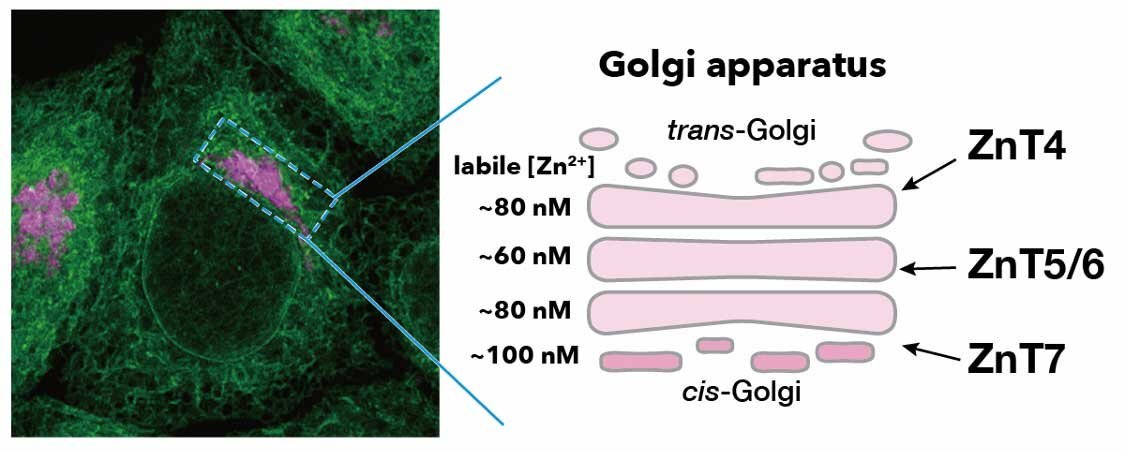
A fluorescence image of the ER and Golgi apparatus. Authors revealed that the Golgi-resident zinc transporters, ZnT4, ZnT5/6, and ZnT7, serve to maintain zinc homeostasis at the different Golgi cisternae. This mechanism finely tunes the localization, traffic and function of the ER-Golgi-cycling chaperone protein, ERp44. ©Yuta Amagai et al.
ERp44 captures immature secretory proteins at the Golgi apparatus to prevent their abnormal secretion. Previous studies have shown that mice with the expression of ERp44 suppressed suffer from heart failure and hypotension.
Additionally, many secretory zinc enzymes are related to various diseases, including metastasis of cancer cells and hypophosphatasia. These enzymes depend on the Golgi-resident ZnT complexes to acquire Zn2+ for enzymatic activity. Male mice with ZnT5 suppressed have experienced death caused by arrhythmias, so there is possible relevance of Zn2+ homeostasis to cardiovascular disease.
"Our findings will help us understand the mechanism by which disruptions of Zn2+ homeostasis in the early secretory pathway leads to the development of pathological conditions," adds Inaba.
The group are hopeful that the strategies employed in their study can paint a bigger picture of the mechanisms underlying the maintenance of intracellular Zn2+ homeostasis, recommending future studies that can measure Zn2+ in other organelles such as the mitochondria and nucleus.
Publication Details:
Title: Zinc homeostasis governed by Golgi-resident ZnT family members regulates ERp44-mediated proteostasis at the ER-Golgi interface
Authors: Y. Amagai, M. Yamada, T. Kowada, T. Watanabe, Y. Du, R. Liu, S. Naramoto, S. Watanabe, J. Kyozuka, T. Anelli, T. Tempio, R. Sitia, S. Mizukami, and K. Inaba
Journal: Nature Communications
DOI: 10.1038/s41467-023-38397-6
Contact:
Kenji Inaba
Institute of Multidisciplinary Research for Advanced Materials
Email: kenji.inaba.a1 * tohoku.ac.jp (Replace * with @)
Website: http://www2.tagen.tohoku.ac.jp/lab/inaba/html/
Posted on:May 29, 2023




